In the pharmaceutical and prescription drug industries, the ability to test and release products safely is foundational. Without validated analytical testing, finished goods cannot ship, supply chains stall, and patient access can be disrupted.
Recently, a prescription drug manufacturer specializing in generic products found itself in a vulnerable position.
The Challenge: Testing Disruption with a Tight Deadline
The manufacturer had developed a unique technology platform to produce its generic drug products. However, they unexpectedly realized they would not be able to conduct the required analytical chemistry testing in-house to support product release as they originally planned.
The implications were significant:
- Product could not be released to customers
- Shipments would halt
- Revenue and patient supply were at risk
- Regulatory compliance had to be maintained
To make matters more challenging, the call to Q Laboratories came just before Thanksgiving, with a hard deadline to be fully operational by mid-January, approximately six weeks.
The Response: Rapid Alignment and Execution
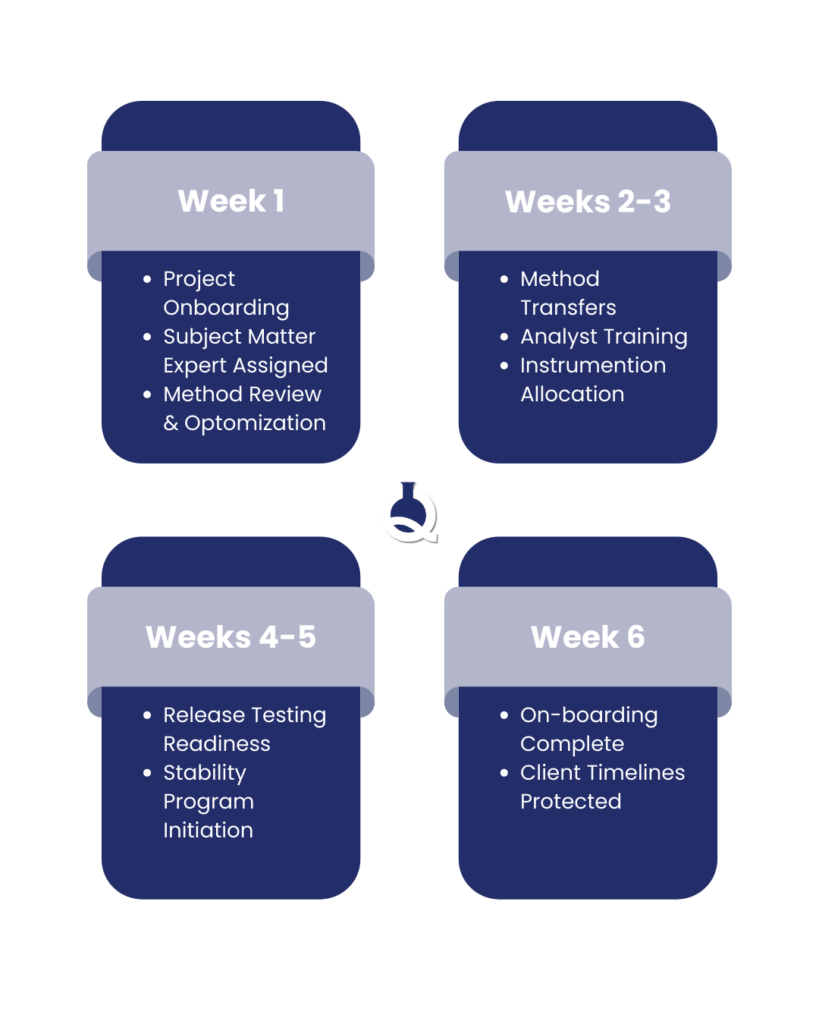
Q Laboratories assembled a dedicated team of scientists and technical experts to support the client.
This was not routine testing support. It required:
- Rapid onboarding and project assessment
- Allocation of dedicated analytical instrumentation
- Full adherence to regulatory requirements
- Method transfers across multiple product categories
- Implementation of stability programs
A Q Laboratories subject matter expert (SME) was immediately assigned to lead the project, providing technical oversight and consistent communication throughout the engagement.
Method Transfer & Optimization
The scope included transferring several analytical chemistry methods covering:
- Raw materials
- Active Pharmaceutical Ingredients (API)
- Finished dosage forms
As is common in method transfer projects, the team conducted a detailed review of each method and, where appropriate, adjusted certain parameters to improve robustness and performance while maintaining compliance and validation requirements.
Multiple analysts were trained on each newly onboarded method to ensure continuity and operational resilience.
Stability Programs Initiated
At the same time, the regulated drug products and dosage strengths were placed on stability programs to support ongoing regulatory and quality commitments. This allowed the client to maintain long-term compliance while addressing immediate release testing needs.
The Outcome: Continuity Maintained
Despite the compressed timeline and holiday timing, the methods were successfully transferred in, testing began, stability studies were initiated, and product release timelines were maintained.
The manufacturer was able to continue shipping product without interruption.
The Role of a Laboratory Partner
For prescription drug and pharmaceutical manufacturers, analytical testing is more than a support function; it directly affects supply continuity and regulatory compliance.
In situations like this, the differentiators are often practical:
- Experienced technical leadership
- Cross-trained analytical teams
- Regulatory-focused processes
- Available capacity
- The ability to move quickly when needed
In this case, six focused weeks of coordinated effort allowed the client to stay on track and avoid disruption.


